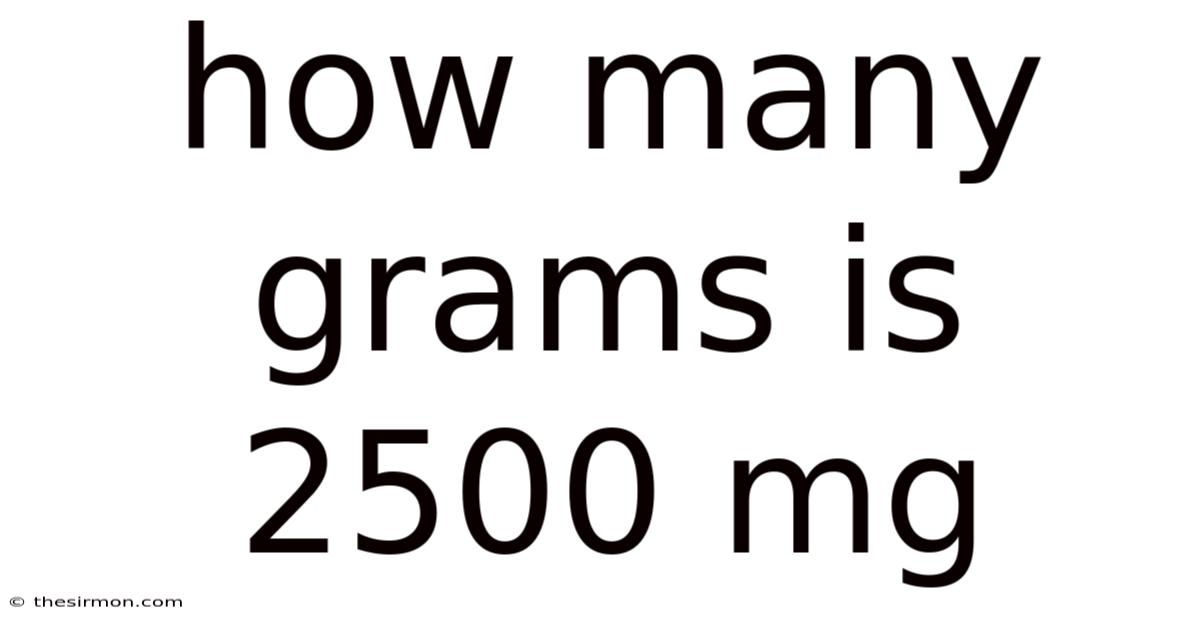The precise conversion between milligrams and grams forms a cornerstone of scientific measurement, underpinning everything from laboratory experiments to everyday cooking. By mastering these principles, users can avoid common pitfalls that might otherwise lead to miscalculations, thereby enhancing precision in their work and decisions. It serves as a foundational resource for anyone seeking to deal with the complexities of unit conversion, whether in academic settings, professional environments, or personal projects. On the flip side, this article delves deeply into the mechanics behind such conversions, exploring not only the mathematical foundations but also the practical implications of grasping these relationships. While many individuals may instinctively rely on informal approximations, understanding exact transformations ensures accuracy in tasks ranging from pharmacy practices to engineering calculations. The process involves recognizing the proportional relationship between the two units and applying appropriate scaling factors to bridge the gap between the smaller magnitude of milligrams and the larger unit of grams. Think about it: such knowledge empowers individuals to solve real-world problems with confidence, ensuring that numerical values remain consistent and reliable across diverse applications. The significance extends beyond mere arithmetic; it reflects a commitment to accuracy, a value that resonates across disciplines and professions alike.
Not obvious, but once you see it — you'll see it everywhere.
Understanding the Basics of Unit Conversion
At the heart of any conversion lies a fundamental principle: ratios between units. Milligrams (mg) and grams (g) represent distinct scales, with one milligram equating to one-thousandth of a gram. This relationship is rooted in the scientific definition of the metric system’s consistency, where smaller units are fractions of larger ones. Here's a good example: 1 gram encompasses 1,000 milligrams, a ratio that underscores the importance of grasping these proportions. When converting from milligrams to grams, one must recognize that multiplying by 1000 reverses the division inherent in the relationship. Conversely, converting grams back to milligrams requires dividing by 1000, a reciprocal operation that demands careful attention to prevent errors. This reciprocal nature highlights the necessity of maintaining clarity between the two units throughout the conversion process. Additionally, contextual factors such as the unit’s application—whether in scientific contexts requiring precision or in culinary measurements where accuracy is very important—dictate which conversion pathway is most appropriate. Understanding these nuances ensures that the chosen method aligns with the specific scenario at hand, preventing misinterpretations that could compromise outcomes. Such awareness also extends to unit consistency within broader frameworks, where maintaining uniformity in terminology and measurement standards becomes critical for coherence and clarity Nothing fancy..
Calculations and Applications in Practice
Applying these principles in practice involves translating real-world scenarios into mathematical operations, where precision is non-negotiable. Consider a scenario where a recipe calls for a precise amount of flour measured in milligrams, requiring conversion to grams for laboratory use. Here, accurate conversion ensures the correct proportions are maintained, preventing potential spoilage or inefficiency. Alternatively, in pharmaceutical contexts, precise dosages depend on accurate milligram-to-gram conversions to avoid underdosing or overdosing patients. The process often necessitates careful calculation, especially when dealing with large numbers or multiple conversions. Here's one way to look at it: converting 2500 milligrams into grams involves dividing by 1000, yielding 2.5 grams—a straightforward calculation that underscores the utility of such skills. Such applications demonstrate how foundational knowledge translates directly into tangible results, reinforcing the value of mastering these techniques. To build on this, the application extends beyond individual tasks; in fields like finance, engineering, or environmental science, precise unit handling can influence outcomes significantly. Whether adjusting material quantities for manufacturing or analyzing data sets, the ability to convert units accurately ensures that decisions are grounded in reliable data. This versatility underscores the universal relevance of unit conversion, making it a skill applicable far beyond the confines of a single discipline No workaround needed..
Common Mistakes and Their Consequences
Despite the clear theoretical framework, errors often arise due to oversight or misapplication of conversion rules. One prevalent mistake involves misapplying the conversion factor, such as incorrectly assuming 1 gram equals 1 milligram rather than recognizing the inverse relationship. Such errors can lead to significant discrepancies, particularly when dealing with large numbers or when dealing with decimal precision. Another pitfall lies in neglecting unit context; for instance, confusing milligrams with micrograms or vice versa, which can result in drastically different outcomes. Additionally, overcomplicating calculations by introducing unnecessary steps can obscure
The interplay between precision and precision defines the foundation of trust in systems where accuracy underpins success. Such alignment ensures that diverse perspectives converge into cohesive outcomes, fostering reliability and consistency. In this context, mastery transcends mere technicality, becoming a cornerstone for sustained progress. Thus, adherence remains indispensable Small thing, real impact..
Continuing from the pointwhere the previous paragraph left off, it becomes evident that the most insidious errors stem from a lack of systematic verification. When a conversion is performed mentally, the absence of a written checkpoint often allows slip‑ups to proliferate unnoticed. Here's a good example: converting 7 500 mg to grams by simply moving the decimal point one place to the left yields 75 g, yet the correct result is 7.5 g. Also, the mistake is not merely arithmetic; it reflects a misunderstanding of the magnitude implied by the prefix “milli‑,” which denotes a factor of one‑thousandth. Recognizing that each step down the metric ladder reduces the numerical value by three orders of magnitude can prevent such oversights Easy to understand, harder to ignore..
Another frequent source of error involves the misuse of conversion tables, particularly when multiple units are stacked in a single problem. Skipping the intermediate step or applying the wrong factor—such as multiplying by 1 000 instead of 1 000 000—produces a result that is off by three orders of magnitude, potentially jeopardizing the experiment’s validity. That's why 045 kg = 45 g) and then grams into micrograms (45 g = 45 000 000 µg). The correct pathway is to first transform kilograms into grams (0.045 kg of a reagent into micrograms. Imagine a chemistry experiment that requires converting 0.The lesson here is that breaking complex conversions into discrete, verifiable stages not only clarifies the process but also creates built‑in error‑checking mechanisms Nothing fancy..
A subtler yet equally damaging oversight is the neglect of significant figures when converting between units. Here's the thing — precision is not an abstract notion; it carries practical weight in fields ranging from aerospace engineering to nutrition labeling. Suppose a dosage calculation yields 0.003 g of an active ingredient. Here's the thing — if the original measurement was recorded to three decimal places in milligrams (3. 000 mg), the converted value should retain the same level of certainty, i.Also, e. Also, , 3. Think about it: 000 mg = 3. 000 g × 10⁻³ = 0.Worth adding: 003000 g. Truncating to 0.003 g without the trailing zeros can mislead a pharmacist into believing the dosage is less precise than it truly is, potentially prompting an unnecessary dosage adjustment. Maintaining the appropriate number of significant figures therefore safeguards both the integrity of the data and the safety of downstream applications Simple, but easy to overlook..
Beyond individual blunders, systemic issues can amplify the impact of unit‑conversion errors. A notable example occurred in a large‑scale construction project where a subcontractor mistakenly interpreted “150 mm” as “150 cm,” resulting in structural components that were ten times larger than designed. Even so, the ensuing redesign not only delayed the schedule but also incurred substantial financial penalties. In collaborative projects, inconsistent documentation of units—some team members using metric prefixes, others relying on imperial equivalents—can lead to misaligned expectations and costly rework. Such incidents underscore the importance of establishing a unified unit‑conversion protocol at the outset of any multidisciplinary effort, including standardized checklists, shared conversion references, and mandatory peer reviews.
To mitigate these pitfalls, several best practices have emerged within professional communities. First, always annotate intermediate results with their units; this visual cue serves as a constant reminder of the physical quantity being manipulated. Now, second, put to work digital calculators or conversion utilities that enforce unit consistency, thereby reducing reliance on manual mental arithmetic. Third, adopt a habit of “reverse‑checking” the answer: after converting 2 500 mg to grams (yielding 2.5 g), multiply back by 1 000 to confirm the original magnitude. Finally, cultivate a culture of questioning—encourage team members to verbalize the rationale behind each conversion step, fostering collective scrutiny and reducing the likelihood of unchecked mistakes.
Simply put, mastering unit conversion is far more than an academic exercise; it is a critical competency that underpins safety, efficiency, and innovation across a multitude of disciplines. So by internalizing the logical structure of the metric system, adhering to rigorous verification techniques, and embedding standardized protocols within collaborative workflows, practitioners can transform a seemingly mundane calculation into a reliable foundation for broader scientific and engineering endeavors. The ability to deal with between milligrams, grams, and their myriad derivatives with confidence not only prevents costly errors but also empowers individuals to translate abstract measurements into concrete, actionable outcomes—ultimately reinforcing the very fabric of precision that drives progress.
Worth pausing on this one.
Conclusion
Accurate unit conversion is the silent engine that propels precise measurement into reliable action. Whether in the laboratory, the clinic, the factory floor, or the boardroom, the disciplined application of conversion principles safeguards against error, enhances decision‑making, and sustains the trust upon which complex systems are built. By embracing systematic verification, respecting significant figures, and fostering collaborative standards, we confirm that every milligram, gram, or larger unit contributes meaningfully to the collective pursuit of accuracy and excellence. In this way, the simple act of converting