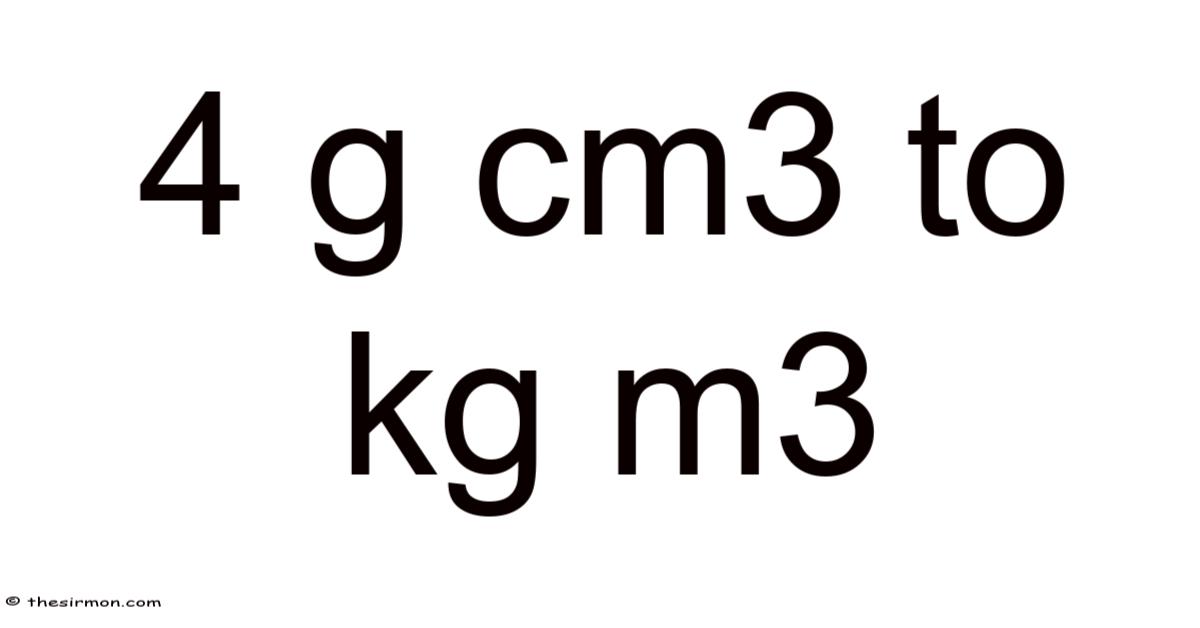Understanding the Conversion: How to Convert 4 g/cm³ to kg/m³
Converting density measurements is a fundamental skill in physics, chemistry, and engineering, and understanding how to transform 4 g/cm³ to kg/m³ is a perfect way to master the logic behind unit analysis. So density, defined as mass per unit volume, is a crucial property that determines whether an object will sink or float, how much material is required for construction, and how substances behave under pressure. While grams per cubic centimeter (g/cm³) is a common unit in laboratory settings, the SI (International System of Units) standard for density is kilograms per cubic meter (kg/m³). This article provides a deep dive into the mathematical process, the scientific reasoning, and practical examples to ensure you never struggle with density conversions again Easy to understand, harder to ignore..
You'll probably want to bookmark this section.
What is Density? A Scientific Overview
Before diving into the math, You really need to understand what we are actually converting. Density ($\rho$) is a physical property of matter that expresses the relationship between an object's mass and the space it occupies. The formula for density is:
$\text{Density} = \frac{\text{Mass}}{\text{Volume}}$
When we say a substance has a density of 4 g/cm³, we are stating that every single cubic centimeter of that substance weighs exactly 4 grams. To convert this to kg/m³, we are essentially looking for the mass in kilograms contained within a much larger volume: one cubic meter. Because a cubic meter is significantly larger than a cubic centimeter, we expect the numerical value of the density to increase significantly during the conversion Small thing, real impact..
The Step-by-Step Mathematical Conversion
To convert 4 g/cm³ to kg/m³ accurately, we must perform a two-step conversion: one for the mass (grams to kilograms) and one for the volume (cubic centimeters to cubic meters). Using the factor-label method (also known as dimensional analysis) is the most reliable way to ensure accuracy.
Step 1: Convert Mass from Grams (g) to Kilograms (kg)
We know that the prefix "kilo" means 1,000. Therefore:
- $1 \text{ kg} = 1,000 \text{ g}$
- To convert grams to kilograms, we must divide by 1,000 or multiply by the fraction $\frac{1 \text{ kg}}{1,000 \text{ g}}$.
Step 2: Convert Volume from Cubic Centimeters (cm³) to Cubic Meters (m³)
This is where most students make mistakes. We know that:
- $1 \text{ m} = 100 \text{ cm}$
On the flip side, because we are dealing with volume (three dimensions), we must cube the entire conversion factor:
- $(1 \text{ m})^3 = (100 \text{ cm})^3$
- $1 \text{ m}^3 = 1,000,000 \text{ cm}^3$ (or $10^6 \text{ cm}^3$)
To convert $\text{cm}^3$ to $\text{m}^3$, we must divide by 1,000,000 or multiply by the fraction $\frac{1 \text{ m}^3}{1,000,000 \text{ cm}^3}$ Practical, not theoretical..
Step 3: Combine the Conversions
Now, let's put it all together for our specific value of 4 g/cm³:
$\text{Density} = 4 \frac{\text{g}}{\text{cm}^3} \times \left( \frac{1 \text{ kg}}{1,000 \text{ g}} \right) \times \left( \frac{1,000,000 \text{ cm}^3}{1 \text{ m}^3} \right)$
Let's simplify the math:
- First, multiply the numerators: $4 \times 1 \times 1,000,000 = 4,000,000$
- Next, multiply the denominators: $1,000 \times 1 = 1,000$
Final Result: 4 g/cm³ is equal to 4,000 kg/m³.
The "Quick Rule" for Density Conversion
If you are working on a timed exam or a quick calculation in a lab, you might not want to write out the full dimensional analysis every time. There is a mathematical shortcut you can memorize:
To convert from g/cm³ to kg/m³, simply multiply the value by 1,000.
Example:
- $1 \text{ g/cm}^3 = 1,000 \text{ kg/m}^3$
- $2.5 \text{ g/cm}^3 = 2,500 \text{ kg/m}^3$
- $4 \text{ g/cm}^3 = 4,000 \text{ kg/m}^3$
Conversely, if you need to go from the larger unit (kg/m³) to the smaller unit (g/cm³), you simply divide by 1,000 That's the part that actually makes a difference. No workaround needed..
Why Does the Number Get So Much Larger?
It can feel counterintuitive that the number jumps from 4 to 4,000. This happens because of the massive difference in scale between the units And that's really what it comes down to. Practical, not theoretical..
Imagine a small cube the size of a sugar cube (1 cm³). It weighs 4 grams. Now, imagine a massive cube that is 1 meter wide, 1 meter long, and 1 meter high (1 m³). That giant cube is made up of one million tiny sugar-cube-sized spaces. If every single one of those million spaces contains 4 grams of material, the total mass becomes enormous. When we convert that total mass into kilograms, we end up with 4,000 kg Worth knowing..
Practical Applications of Density Conversion
Understanding these conversions is not just an academic exercise; it is vital in various professional fields:
- Marine Engineering: When calculating the buoyancy of a ship, engineers must know the density of seawater (approximately $1,025 \text{ kg/m}^3$) versus the density of the ship's hull materials.
- Geology and Mining: Geologists identify minerals by their density. A density of $4 \text{ g/cm}^3$ might indicate a specific type of heavy metal or silicate rock.
- Material Science: When manufacturing polymers or alloys, scientists use $\text{g/cm}^3$ in small-scale lab tests but must report findings in $\text{kg/m}^3$ for industrial-scale production and shipping logistics.
- Meteorology: The density of air changes with altitude and temperature. While air density is quite low, precise conversions are necessary for flight calculations and weather modeling.
Frequently Asked Questions (FAQ)
1. Is 4 g/cm³ a high density?
Yes, compared to water, which has a density of $1 \text{ g/cm}^3$ (or $1,000 \text{ kg/m}^3$), a substance with $4 \text{ g/cm}^3$ is four times as dense. It would sink rapidly in water. Common materials with densities in this range include certain types of heavy stones or metal alloys Easy to understand, harder to ignore..
2. Why do we use two different units for density?
$\text{g/cm}^3$ is very convenient for small objects, like a gemstone or a small sample of liquid in a test tube, because the numbers remain small and easy to manage. $\text{kg/m}^3$ is the standard SI unit used in scientific formulas and large-scale engineering because it integrates smoothly with other SI units like Newtons (force) and Pascals (pressure) And that's really what it comes down to..
3. Can I just multiply by 100 to convert?
No. A common error is to assume that because there are 100 cm in a meter, you only multiply by
3. Can I just multiply by 100 to convert?
No. A common error is to assume that because there are 100 cm in a meter, you only multiply by 100. That said, density involves cubic units. Since 1 meter equals 100 centimeters, 1 cubic meter equals 100³, or 1,000,000 cubic centimeters. So, you must multiply by 1,000 to convert from g/cm³ to kg/m³ And that's really what it comes down to..
4. What happens if I confuse the units?
Mixing up these units can lead to significant miscalculations. Take this: if you're designing a bridge and mistakenly use 4 g/cm³ instead of 4,000 kg/m³ in your structural calculations, your safety margins could be off by a factor of 1,000. In aerospace engineering, such an error could be catastrophic when calculating material requirements or fuel efficiency Practical, not theoretical..
5. How do temperature and pressure affect density?
Temperature and pressure can significantly impact density measurements. Most materials expand when heated and contract when cooled, which changes their volume and thus their density. Similarly, increasing pressure typically decreases volume, increasing density. Scientists often specify conditions (usually at standard temperature and pressure, or STP) when reporting densities to ensure accuracy.
Key Takeaways
The conversion from grams per cubic centimeter to kilograms per cubic meter isn't just about moving decimal places—it's about understanding the fundamental relationship between mass and volume at different scales. Here are the essential points to remember:
- 1 g/cm³ = 1,000 kg/m³ — this is your baseline conversion factor
- Always consider the cubic relationship when dealing with volume conversions
- Small numbers in g/cm³ become large numbers in kg/m³ due to the million-fold volume difference
- Context matters: use g/cm³ for small-scale work and kg/m³ for engineering and scientific calculations
Conclusion
Mastering density conversions is more than memorizing a mathematical operation—it's about developing an intuitive understanding of how matter behaves at different scales. Whether you're a student working through textbook problems or a professional engineer designing critical infrastructure, the ability to easily convert between g/cm³ and kg/m³ ensures accuracy in your work and prevents potentially costly mistakes.
The next time you encounter a density measurement, remember that behind every number lies a story about the material's composition, behavior, and suitability for its intended application. By appreciating both the mathematical relationships and their practical implications, you'll be better equipped to make informed decisions in science, engineering, and everyday problem-solving And that's really what it comes down to..
Counterintuitive, but true.